"My Ferritin is Low, Should I Supplement Iron?"
Is It Low Iron, Hypothyroidism, Liver Dysfunction or Low Copper? Clearing Up the Iron Confusion
Disclaimer: Content for entertainment purposes only. Not medical or health advice.
Next to magnesium, iron is arguably the most life-sustaining metal. Iron is used to make hemoglobin, the oxygen-delivering protein that ensures our cells’ survival.
Without oxygen, the cells making up our tissues suffocate, as they can’t make the energy they rely on to perform vital functions, maintain their structure, communicate with one another, and repair damage. Various levels of cellular energy deficiency are involved in all health struggles.
Since a true iron deficiency impairs cells’ ability to make energy, the list of iron deficiency symptoms is long. It includes fatigue, breathlessness, hair loss, brittle nails, weakness, insomnia, cold hands and feet, chest pain, depression, irritability, difficulty concentrating, restless leg syndrome, exercise intolerance, and more. Every organ, every cell, and every function takes a hit when iron (and thus cellular energy) is in short supply.
Table of Contents:
Iron Deficiency Symptoms are Indistinguishable from “Energy Deficiency” Symptoms
Iron Deficiency vs. Trapped Iron: Spotting the Difference & Remediation Strategies
The Iron Recycling System & Microbiome Disruption: Why I’m So Cautious With Iron Supplements
Iron Deficiency Symptoms are Indistinguishable from “Energy Deficiency” Symptoms
Seeing the symptoms of iron deficiency, you may be thinking that these symptoms overlap almost entirely with the symptoms of hypothyroidism, or with magnesium deficiency symptoms, or even with the symptoms of being deficient in the B vitamins. This is because, at the core, they’re all symptoms of cellular energy deficiency.
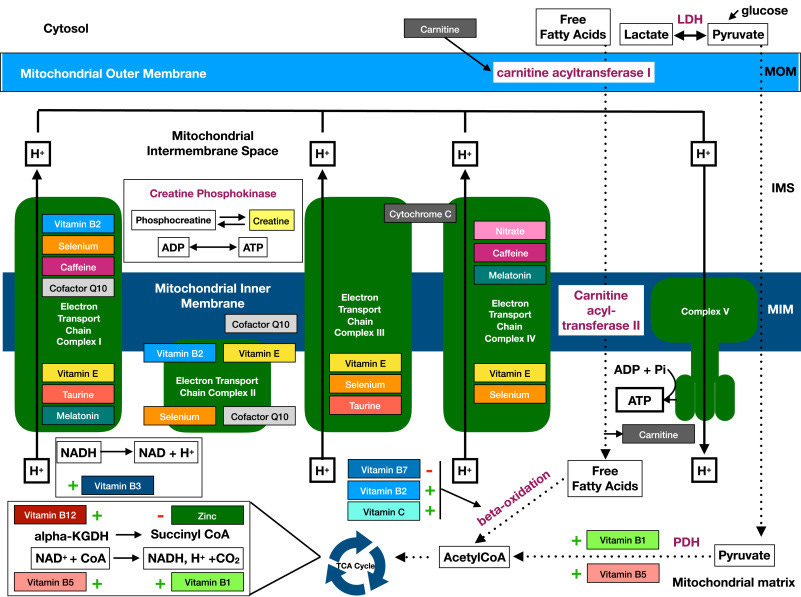
It takes more than oxygen (supplied to cells by iron-containing hemoglobin) for cells to make energy. To run the energy creation “assembly line” you need oxygen, but also certain vitamins (incl. vitamins B1, B2, B3, B5, B7, C and E), minerals (incl. magnesium, zinc, copper, and iron), Coenzyme Q10, adequate thyroid hormone concentrations, adequate protein and, most importantly, adequate fuel, in the form of glucose and fats.
Hormones, neurotransmitters, immune cytokines, antioxidants, drugs, environmental toxins, and other vitamins and minerals also affect this entire energy-making cascade, but largely in more indirect ways. Instead of actually working the “assembly line,” most other compounds simply “tell” certain workstations to work faster or slower, or affect cell communication.
Iron, in addition to powering hemoglobin, plays other roles in this cascade. It’s needed for the creation of thyroid hormones, which dictate how fast or slow energy is made. It’s also a structural component of some of the mitochondrial complexes involved in energy production. In other words, iron is also “employed” at some of the workstations making up the energy creation “assembly line.” It’s a very important metal.
So why is iron, and iron supplementation specifically, so controversial?
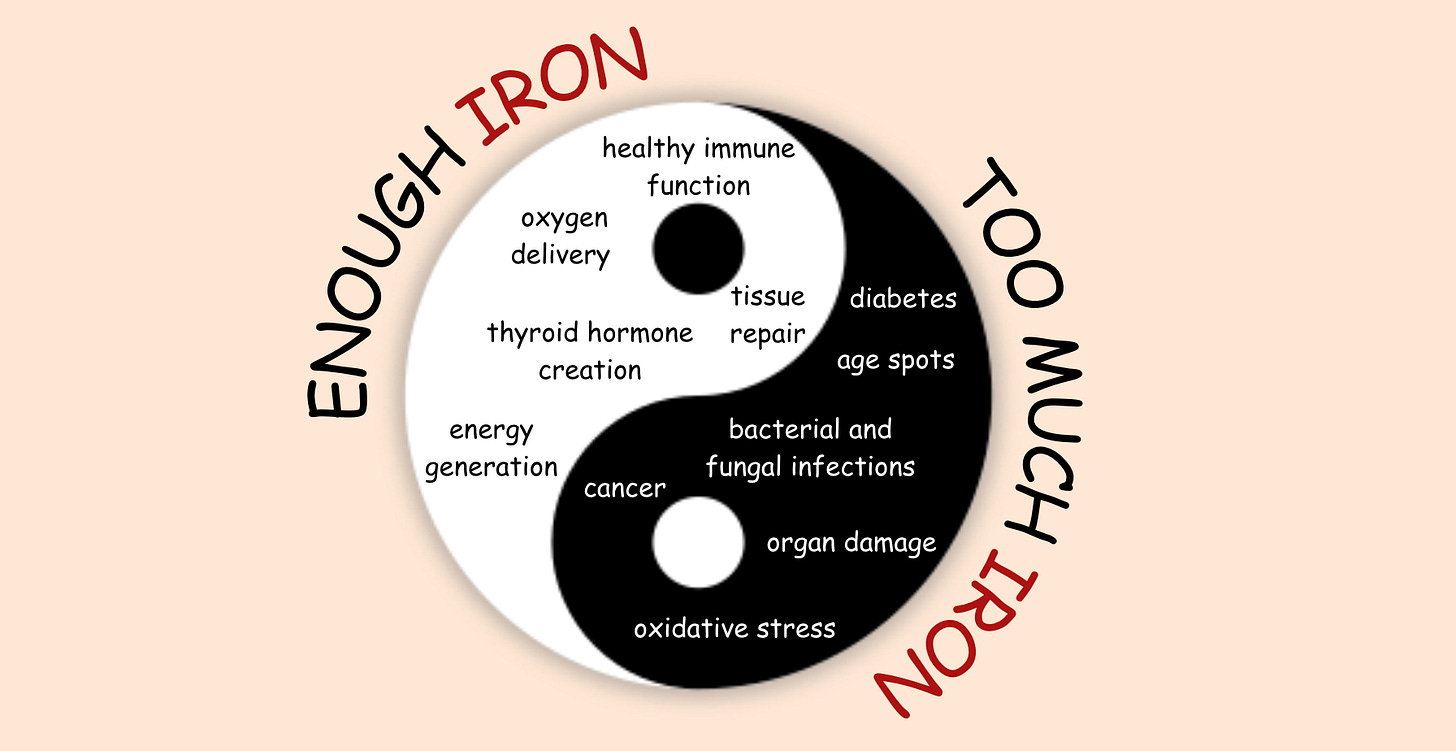
It’s controversial because the very same properties that make iron life-sustaining also make it deadly when in excess. What worsens the problem is that the body lacks ways to rid itself of excess iron. Instead of supporting health, excess iron starts corroding us from the inside.
The Duality of Iron: Essential but Risky
“The body has no mechanism for the active excretion of iron, so body iron levels are controlled at the point of absorption in the small intestine. Disturbances in this equilibrium, such as those leading to enhanced absorption, can have significant clinical consequences. Continued excessive iron uptake is followed by iron deposition in various tissues, ultimately leading to tissue damage, and possibly end-organ failure.”
- from: Anderson GJ. Mechanisms of iron loading and toxicity. Am J Hematol. 2007 Dec;82(12 Suppl):1128-31. doi: 10.1002/ajh.21075. PMID: 17963252.
To understand the duality of iron and why it’s so precious yet dangerous, we need to understand its relationship to oxygen.
Oxygen is what keeps us alive here on planet Earth. Yet, one can argue that oxygen is also what leads to our eventual demise and death.
To stay alive and keep our organs functioning, we need to make cellular energy. This process relies on getting oxygen into cells and then breaking down fuel (carbs, fats) into electrons. These electrons are then handed to oxygen in a controlled manner to combust and generate energy. This is oxidative metabolism. This is the “assembly line.” This is what keeps us alive.
Yet, when the guardrails keeping this “assembly line” intact become shaky, electrons can slip out. When they do, they can react with oxygen unsupervised, outside of the “controlled combustion pipeline.” This is how reactive oxygen species are made. These reactive oxygen species then steal electrons from other molecules, damaging them. This includes membrane fats, DNA, mitochondrial proteins, and anything else that makes up our bodies. These damaged molecules become sources of damage themselves. It becomes a chain reaction, unless an antioxidant steps in to break the chain. This cumulative, sustained damage is one theory behind why we age and eventually succumb to “father time.”
Iron was “chosen” for the job of oxygen delivery because of how reactive it is.
It can very easily grab and let go of electrons, and it can very easily grab and let go of oxygen.
Like an arcade claw machine, it can rapidly grab and drop oxygen wherever it’s needed. Other, more stable metals would not be suited for the role of oxygen delivery as they’d be more likely to grab onto oxygen and not let it go.
Yet, the same properties that make iron the ideal metal for delivering oxygen in animals with high metabolic rates and massive oxygen demands (like us, humans) are what make it dangerous.
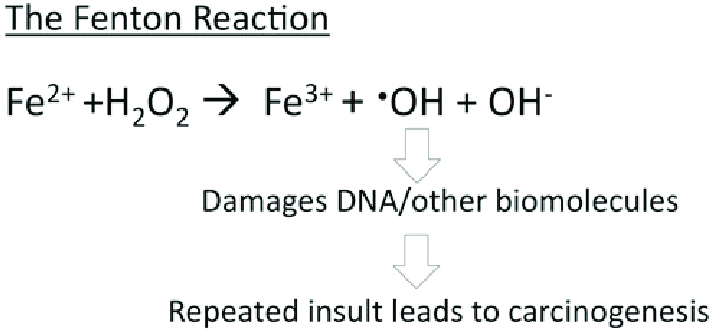
Since iron is so “shaky” and reactive, if it encounters a damaging reactive oxygen species, it easily allows for its electrons to be stolen. This creates the most reactive and damaging reactive oxygen species known - a hydroxyl radical (⋅𝑂𝐻)
This process is called “The Fenton Reaction,” and it occurs specifically when iron reacts with hydrogen peroxide, a reactive oxygen species.
This reactivity and how easy it is for iron to “turn to the dark side” and go rogue is why iron overload can lead to such disparate disease states.
When excess iron gets deposited in the liver or the heart (which are the organs most likely to store it), it can continuously drive oxidative damage, injuring those organs. This is why iron overload is linked to liver failure and heart disease. Other organs are not immune to iron overload, either.
Excess iron’s contribution to oxidative stress fuels metabolic disorders like cancer and diabetes. This is because rampant oxidative stress disturbs the energy creation pipeline, causing a “cellular energy deficiency.” Cells lose the ability to “feed,” communicate with one another, and respond to damage. The pancreas becoming iron-overloaded can contribute to diabetes, too.
Since iron is so indispensable for oxygen delivery and energy production, other organisms living in our bodies (like bacteria, fungi, and parasites) fight hard for it. To our body’s “critter” ecosystem, iron is the equivalent of “the spice” in the Dune universe.
Our body has mechanisms to “hide” iron during active infection for this very reason - to starve pathogens.
Yet, as you may have already guessed, when iron is excessive, pathogens get a free lunch. Trials administering iron to malnourished children in Africa and Asia resulted in increased rates of hospitalizations for and deaths from malaria, salmonella, E.coli, Staph and more.1
“Even modest oral supplements with highly soluble (non-physiological) iron, as typically used in low-income settings, could promote bacteremia by accelerating early phase bacterial growth prior to the induction of immune defenses.”2
While adequate iron is needed to mount an appropriate immune response and respond to damage, this reverses once iron becomes overabundant. Immune defences tank, and the vulnerability to bacteria is further enhanced when the spleen becomes overloaded with iron. Even certain autoimmune disorders are emerging to have iron overload as part of the “root cause.” 3
Excess iron deposited in joints can lead to arthritis. Its deposition in endocrine glands (like the ovaries, the thyroid gland, or the testes) can lead to a whole plethora of hormonal and reproductive problems. Excess iron in the brain is linked to neurodegeneration and dementia. And when iron mixes with unsaturated fats in the body, it can turn into a sort of cellular “gunk” called lipofuscin. Lipofuscin is what makes up “age pigment,” or “liver spots,” but its negatives don’t end at just looking unpleasant. Lipofuscin is the type of cellular garbage that the body struggles to remove, and while lodged in tissues, it suffocates their energy-making potential. While lipofuscin in the skin is most visible, it also deposits in the brain and vital organs. There, it constantly starves tissues of energy and drives oxidative stress.
Speaking of PUFAs (polyunsaturated fats), iron damages unsaturated fats,4 turning them into harmful lipid peroxides. It’s a mechanism unrelated to hydrogen peroxide or hydroxyl radicals. Iron itself damages cellular components that contain polyunsaturated fats. A body full of polyunsaturated fats (omega-3 and 6 fats) is very prone to iron-induced damage.
A Slow Metabolism Makes Iron More Dangerous
A somewhat counterintuitive phenomenon is that oxidative stress tends to increase when the metabolic rate is low.
Reactive oxygen species are naturally produced whenever the “assembly line” is running. This has led many in the past to theorize that slowing the metabolism (slowing our “assembly lines”) would lower the production of reactive oxygen species and extend lifespan. The idea that the slower you run your internal machinery, the longer you’ll live still has strong adherents even today, which is why fasting or caloric restriction are still touted as “life extending.” This is despite the fact that this hypothesis was mostly rooted in our understanding of machines following the “Industrial Revolution” and not biology. “The less you drive your car, the longer it’ll last” doesn’t apply to living beings.
As it so happens, the slower that our “controlled combustion pipelines” move, the easier it is for electrons to slip out and react with oxygen unsupervised.56 The literal opposite of what would intuitively make sense.
Whenever a rogue electron reacts with oxygen, they create a compound called superoxide. The body then deploys an antioxidant enzyme called superoxide dismutase, which turns superoxide to hydrogen peroxide. Hydrogen peroxide is usually the less damaging of the two…unless it encounters iron.
Something that this article by Keith Littlewood of BalancedBodyMind reminded me of is that the expression of superoxide dismutase increases in states of impaired cellular respiration, in response to the increases in oxidative stress following reductive stress. For example, superoxide dismutase levels increase in diabetes.78
To put this in “human terms,” when your oxidative metabolism is slow, there are more chances for electrons to slip out and react with oxygen unsupervised. This results in the creation of a damaging reactive oxygen species called “superoxide.” The body then deploys an antioxidant called “superoxide dismutase” to neutralize superoxide. This antioxidant enzyme turns superoxide into hydrogen peroxide. Hydrogen peroxide is normally less harmful than superoxide. Unfortunately, if hydrogen peroxide encounters iron, it turns into the worst reactive oxygen species known to man.
A slow metabolism = “clogged assembly line” (reductive stress) = more electron slippage = more electrons reacting with oxygen unsupervised = superoxide ↑ (oxidative stress) = superoxide dismutase ↑ = hydrogen peroxide ↑.
Hydrogen peroxide + iron = a hydroxyl radical (⋅𝑂𝐻) 👹
Most Health Practitioners Jump to Iron Supplements Too Soon
Iron supplements are one of the most commonly purchased supplements. Most women I meet supplement iron. Yet, knowing its potential “dark side,” iron supplements should not be handed out haphazardly.
Don’t get me wrong, there is absolutely such a thing as an iron deficiency. You definitely might need more iron. Iron is absolutely essential, even if an excess of it can cause havoc. It is entirely possible that iron is your missing piece and the reason why you’re feeling cold, dizzy and fatigued.
However, considering how volatile this metal is, I find it crucial to make sure that you are really, really, for sure deficient instead of immediately throwing iron supplements at the issue, as most do.
What it really comes down to is:
Making sure that iron is definitely deficient…
…And increasing iron in ways that are safe and not excessive.
Unfortunately, most healthcare professionals do neither.
The first error that’s often committed by conventional and alternative health practitioners alike is equating anemia to iron deficiency.
Anemia is Not Synonymous With Iron Deficiency
“While iron deficiency is regarded as the major cause of nutritional anaemia, changes in vitamins A, B12, C and E, folic acid and riboflavin status have also been linked to its development.”
- from: Fishman SM, Christian P, West KP. The role of vitamins in the prevention and control of anaemia. Public Health Nutr. 2000 Jun;3(2):125-50. PMID: 10948381.
Anemia and iron deficiency are two different things.
Anemia is characterized by low hemoglobin levels and low hematocrit (the percentage of red blood cells in your blood).
By definition, anemia is a state of hemoglobin and red blood cell deficiency.
It takes way more than iron to make hemoglobin and ensure the survival and creation of red blood cells. Many things can impair hemoglobin or red blood cell creation, ones entirely unrelated to iron.
Anemia can be caused by:
The deficiency of any one of the B vitamins, especially B1, B2, B6, B9 and B12. All of these are involved in hemoglobin creation and red blood cell survival.
The deficiency of fat-soluble vitamins A, D, and E. Vitamin E gets damaged when eaten together with iron, and iron overload can deplete vitamin E by increasing oxidative stress.
“Vitamin E deficiency can also cause anemia because erythrocytes are more susceptible to hemolysis.”9
The deficiency of certain minerals, especially magnesium, selenium, zinc and copper. They play roles in iron mobilization, hemoglobin synthesis, or guarding red blood cells from damage.
High estrogen levels, as they suppress the bone marrow. I covered this in my article “How Hormonal Birth Control Causes Anemia.”
Low protein diets, general malnutrition, and under-eating.
Hypothyroidism, which suppresses the bone marrow.
“Normal levels of thyroid hormones play a role in hemoglobin (Hb) synthesis in adults, and low levels of thyroid hormones affect the hematopoietic process, leading to anemia by slowing oxygenation. […] In up to 25% of patients with hypothyroidism, anemia normalizes with T4 replacement, even in the presence of normal iron, vitamin B12, and folate levels.”10
Infections, which can suppress the bone marrow.
The accumulation of toxic metals, like lead, arsenic, aluminum, nickel and mercury. They either suppress the bone marrow, interfere with steps involved in hemoglobin synthesis, or accelerate the rate at which red blood cells die.
“Lead poisoning has also been found to be the cause of anaemia in a number of cases as lead inhibits porphobilinogen synthase and ferrochelatase, preventing both porphobilinogen formation and the incorporation of iron into protoporphyrin IX, which prevents heme synthesis or causes ineffective heme synthesis and subsequently microcytic anaemia.”11
Can iron deficiency be the cause of anemia? Absolutely. But it is far from the only cause.
Still, time and time again, I see doctors prescribing iron for anemia without investigating other potential causes.
Some “alternative” doctors might diagnose someone as anemic and “needing iron” simply based on a physical exam, such as them feeling cold, looking pale, losing hair, and being constantly fatigued.
I know far too many women who are extremely anemic, and who are also very clearly hypothyroid due to low-calorie, nutrient-poor diets, who already take iron with zero positive effect on their anemia.
I have elderly family members who have extreme anemia due to low-calorie, nutrient-poor diets and poorly-controlled hypothyroidism on T4-only therapy, who’ve been given iron by their primary physician to “treat the anemia,” only for none of their symptoms to improve and for their ferritin to climb to over 300 ng/mL.
Handing out iron pills like they’re candy whenever a person presents as anemic is lazy and ignores the fact that anemia is a disease of general malnutrition and slow metabolism. Throwing iron pills at it is benign at best and extremely dangerous at worst, since excess iron can further drive the inflammatory state that’s suppressing the bone marrow and promoting the breakdown of red blood cells in the first place.
It’s possible to be anemic and have enough iron. It’s even possible to be anemic and have too much iron, since far more than just iron goes into hemoglobin creation and red blood cell survival. Albeit that second scenario would likely be rare because someone malnourished enough to be anemic would likely not be getting excess iron via diet, unless supplementing.
“Anaemia of inflammation is characterized by ID [iron deficiency] despite adequate iron stores.”12
If someone is dealing with anemia, these are the things I’d focus on before jumping straight to iron supplements:
Adequate caloric intake. At least 2,000+ kcal/day for women and 2,500+ kcal/day for men, split across 3 meals: breakfast, lunch and dinner
At least two of the following animal foods eaten daily: 2 eggs, 1 can of oysters, 100 grams of red meat (beef, lamb or veal), 1 can of sardines (canned in water or olive oil), 50-100 grams of chicken liver (or 30-50 grams of beef liver), 50-100 grams of mussels, 2 glasses of goat milk, 30 grams of Gouda cheese
At least two of the following plant foods eaten daily: 2 oranges, 1 papaya, 1 cherimoya, 1-2 potatoes, 1 sweet potato, 3-4 kiwis, 2-3 prickly pears, 1-2 pomegranates, 10-20 grams of dark chocolate, one cup of thoroughly-cooked kale, 1-2 raw carrots.
Optionally:
1-2 teaspoons of nutritional yeast
A vitamin E supplement, especially if dealing with symptoms indicative of a potential vitamin E deficiency (ovulation pain, bad menstrual cramps, easy bruising, random aches and pains). I would take vitamin E every 2-4 days.
1-2 tablespoons of orange peel marmalade
This would create a nutrient-dense diet that supports optimal thyroid function, hemoglobin creation and red blood cell survival. Calcium, from dairy, helps remove lead from the body, and certain plant foods (esp. pomegranates, carrots, orange peels and dark chocolate) help to create a healthier microbiome.
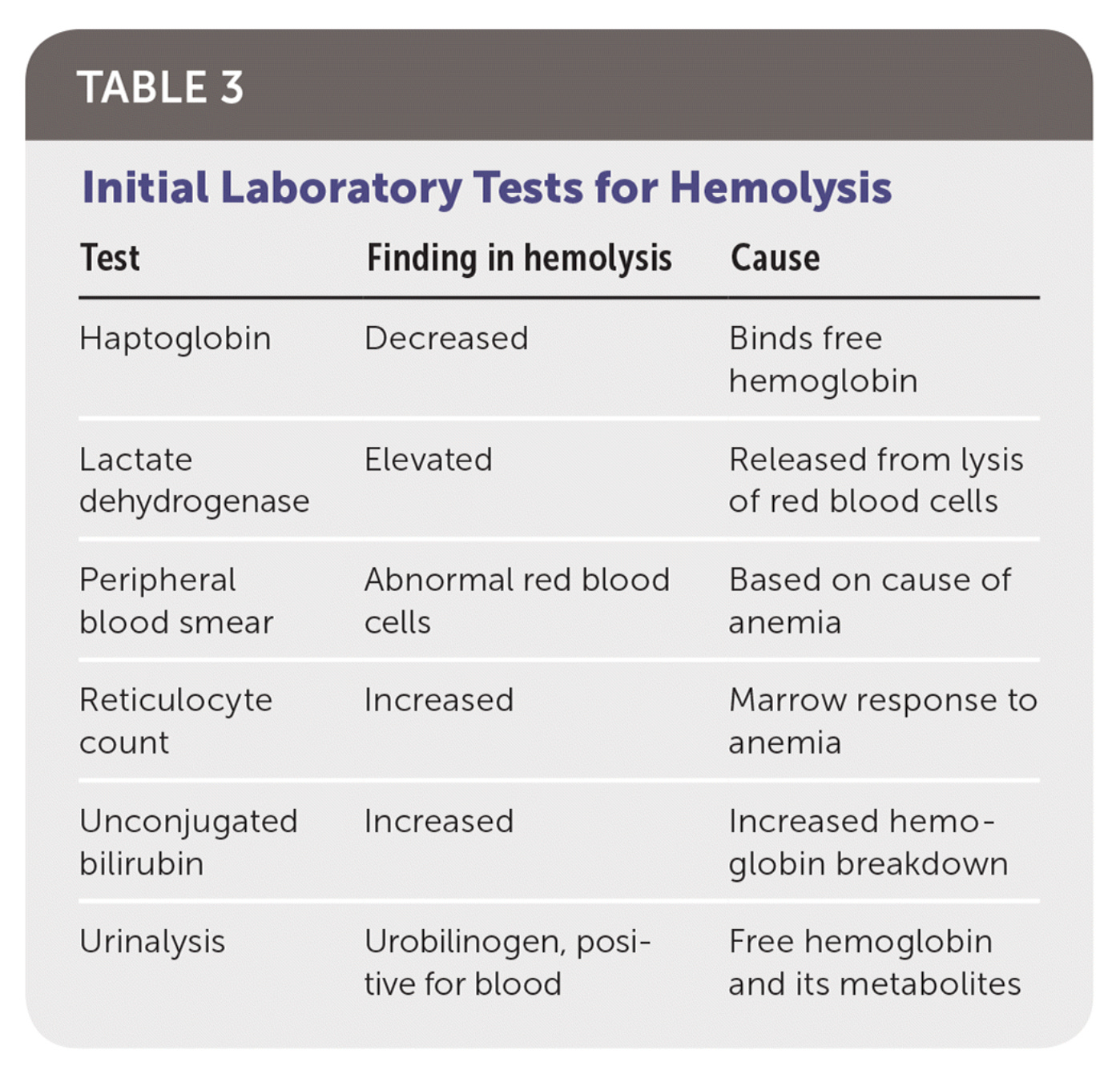
To test for hemolytic anemia, which is anemia caused by red blood cells breaking down faster than they can be replaced, a doctor would normally test:
Complete blood count (CBC)
Reticulocyte count
Haptoglobin
Lactate dehydrogenase (LDH)
Total and indirect bilirubin
The results below would indicate hemolytic anemia, which may be caused by vitamin E deficiency.13
Yet, it is entirely possible that iron is the missing piece.
“How Can I Know If Iron Is My Missing Piece?”
My Substack is a reader-supported publication. You can support my writing and independent research efforts by becoming a paid subscriber. Monthly subscriptions are $15 per month, and can be cancelled at any time. You save with a yearly subscription, as $100 grants you access to my full archive for a year.
Unlock the full version of this article by becoming a paid subscriber.